A Year of Consolidating MPN Research
by David Wallace
 In December 2016, I attended the colossal, premiere hematology event in the world (ASH – American Society of Hematology), in San Diego with a hopeful eye for the 3rd straight year. But there were no ground breaking gene discoveries like 2014, when the Calreticulin (CALR) mutation discovery was announced. I didn’t witness a presentation that carried the level of interest held in the 2015 study “Safety and Efficacy of Combination Therapy of Interferon Alpha2 + JAK 1-2 Inhibitor in the Philadelphia-Negative Chronic MPNs” (click the left arrow to see all 9 slides). During that memorable event, many of the top MPN specialists were there, the lights darkened to a quiet hush, cameras/phones and tablets hovered to capture every slide with nervous anticipation. Not a soul got up and left during the presentation – it’s common place for attendees to shuffle in and out normally.
In December 2016, I attended the colossal, premiere hematology event in the world (ASH – American Society of Hematology), in San Diego with a hopeful eye for the 3rd straight year. But there were no ground breaking gene discoveries like 2014, when the Calreticulin (CALR) mutation discovery was announced. I didn’t witness a presentation that carried the level of interest held in the 2015 study “Safety and Efficacy of Combination Therapy of Interferon Alpha2 + JAK 1-2 Inhibitor in the Philadelphia-Negative Chronic MPNs” (click the left arrow to see all 9 slides). During that memorable event, many of the top MPN specialists were there, the lights darkened to a quiet hush, cameras/phones and tablets hovered to capture every slide with nervous anticipation. Not a soul got up and left during the presentation – it’s common place for attendees to shuffle in and out normally.
There may have been presentations that had attendees glued to their chairs until the end, but as a one man show, I did not witness anything of that magnitude.
Stories Covered
I covered (3) stories that I found noteworthy, there were several others:
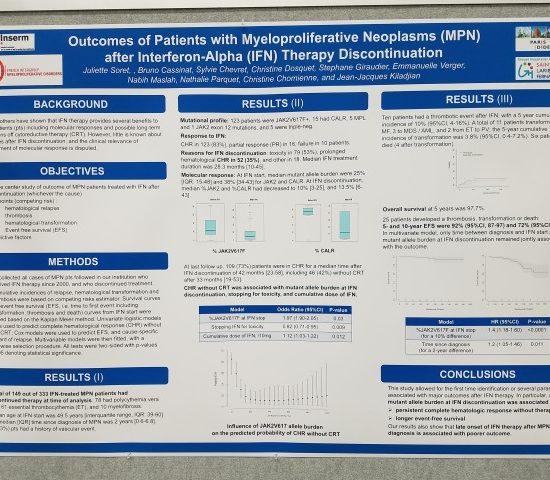
Dr Kiladjian at ASH 2016 Reviews Outcomes of MPN Patients after Interferon therapy discontinuation
Dr Rampal shares MPN updates on Clinical Research with PV Reporter
Interview with Dr Heinz Gisslinger on final results from the PROUD-PV study, A Randomized Controlled Phase 3 Trial Comparing Ropeginterferon Alfa-2b to Hydroxyurea in PV patients.
Interview with Dr Heinz Gisslinger
Due to a scheduling snafu, my previously reserved interview room was given to another reporter. Dr Gisslinger and I were booted to the noisy convention halls, which were not conducive to my very basic “consumer video equipment,” a Sony handycam with a built-in mic and the soft-spoken doctor. So I will summarize a few highlights from the interview. It should be noted PV Reporter covered Dr Gisslinger’s work at ASH 2014, see that video here.
Ropeginterferon (P-1101) was generally equivalent (“not inferior”) to HU in the first year of treatment. P-1101 allows for prolonged intervals of dosing, every 2 to 4 weeks vs daily pills or weekly injections with Pegasys (the interferon currently used in the U.S. and around the world). Interferon is a slow acting agent for long term treatment, while HU (hydroxyurea) causes almost immediate impact on a patient’s blood counts.
Surprisingly, the tolerability of Interferon was superior to HU. The hematological response of Interferon treated patients for 1 year was the same as HU, although HU patients needed dosing reductions due to side effects more frequently than Interferon patients. Typically Interferon related specific side effects, development of auto-immune disease or psychiatric disorders did not occur. Interferon should be started in the early stages of PV
We should look for EU (European Union) approval sometime in 2017 and U.S. FDA approval should follow relatively soon thereafter.
For additional coverage, check out detailed reports from MPN Research Foundation, MPNforum and Patient Power (you will find numerous videos on ASH 2016).
“And now for something completely different,” to borrow a line from Monty Python, we present: